Heart muscle cells (cardiomyocytes) can undergo physiologic and pathologic remodeling when faced with differing hemodynamic loads and neurohormonal environments.
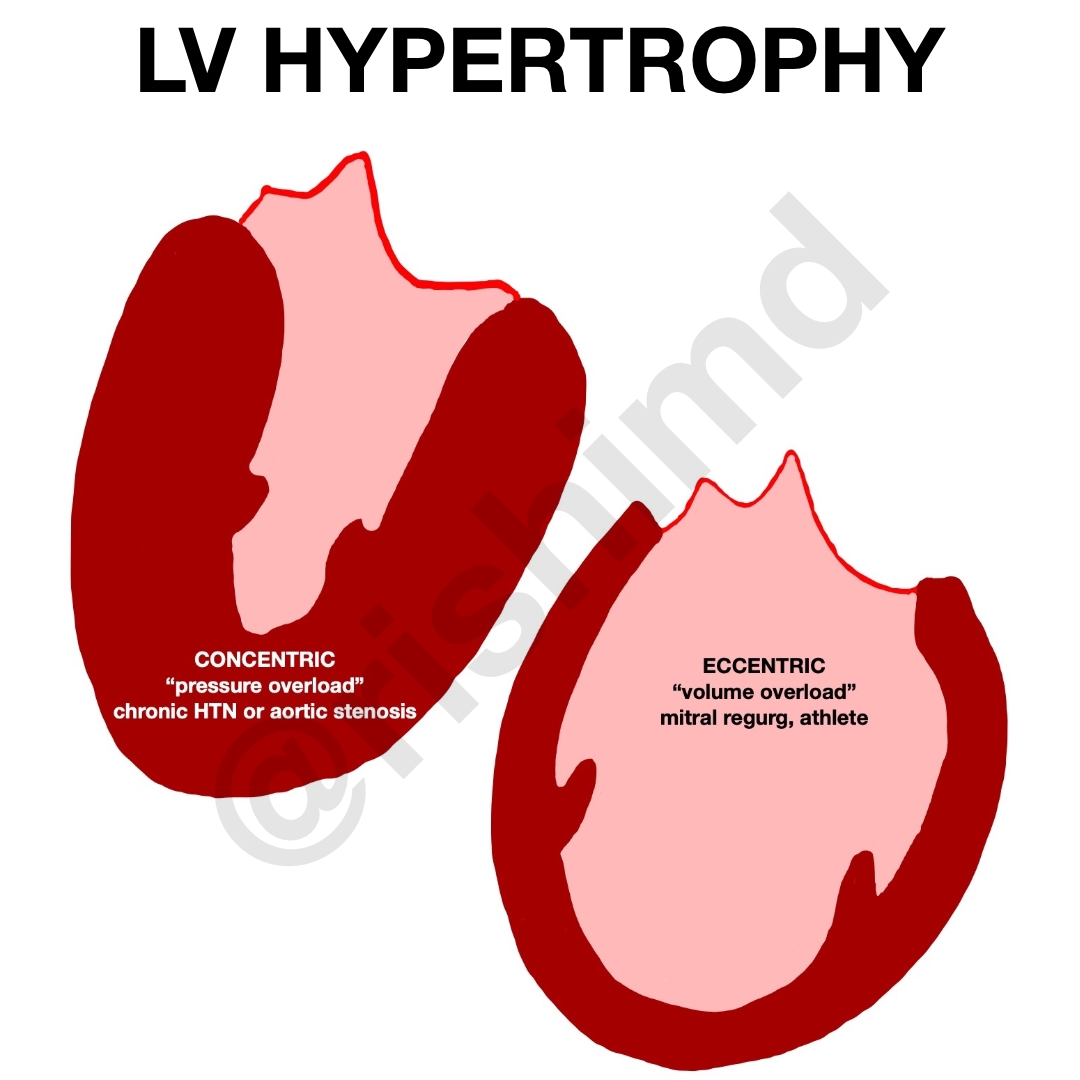
Remodeling occurs in both physiologic and pathologic states. For example, in the conditioned athlete, increased stroke volumes can cause volume overload elongating cardiomyocytes, and leading to the addition of sarcomeres in series. This is also seen in mitral regurgitation due to the relative increase in left ventricular volumes. This eccentric hypertrophy creates a dilated ventricular chamber with relative wall thinning.
In contrast, concentric hypertrophy is more often associated with chronic pressure overload states like hypertension or aortic stenosis. Sarcomeres are added in parallel and cardiomyocytes tend to grow in width leading to thicker walls with smaller chamber sizes. Cardiomyocytes are often replaced with fibroblasts and a collagen network leading to apoptosis of the contractile cells, a stiffer ventricle with increased filling pressures (diastolic dysfunction), and heart failure.
So how does this affect oxygen consumption? It’s a known fact that wall tension is related to myocardial oxygen consumption. This wall tension is directly proportional to intraventricular pressure and chamber radius but inversely proportional to the wall thickness as given by the Law of Laplace:
wall stress = (pressure x radius) / (2 x thickness)
One can see that the larger ventricular thickness in concentric hypertrophy actually creates less wall stress per unit mass compared to the dilated chamber in eccentric hypertrophy. This is because the additional wall stress is spread over a larger ventricular mass compared to a normal baseline. It’s important to understand how to use this knowledge to improve the oxygen supply/demand issues that drive angina pectoris and overall myocardial ischemia.






Dr. Rishi,
Thank you for an excellent and concise article defining the characteristics of eccentric and concentric myocardial hypertrophy. As a certified fitness professional I am aware the “athletes” often used to explain the concentric remodeling associated with sport are runners, cyclists, and sometimes swimmers. These activities are primarily aerobic providing little opportunity to impose acute hemodynamic pressure overload experience with chronic strength training. Moreover, in combination sport and fitness training subjects are exposed to both significant aerobic modifiers which would result in eccentric hypertrophy and anaerobic modifiers which would result in acute bouts of pressure overload and a significantly elevated androgenic hormonal environment all working together to induce concentric remodeling. This is significant because in a natural course of work or activities of daily living the heart is exposed to a combination of aerobic and anaerobic modifiers that should result in myocardial remodeling that presents with both series and parallel sarcomere adaptations. I believe this is of significant importance when using exercise to improve or maintained the heart health and fitness of individuals as they grow older; I am a NASM Senior Fitness Specialist. I would be interested in knowing your opinion on this, especially with regard to the ExerciseisMedicine.org initiative.
Thank you,
Richard Parker, Jr. MS, NASM