Epinephrine (adrenaline). Norepinephrine (noradrenaline). Isoproterenol. Vasopressin. Dopamine. Phenylephrine. Dobutamine. Milrinone. Calcium.
These are just some of the most commonly used vasoactive and inotropic agents to modulate systemic/pulmonary vascular resistance, cardiac inotropy, chronotropy, and the like. But how does one combine these medications to achieve a physiologic endpoint?
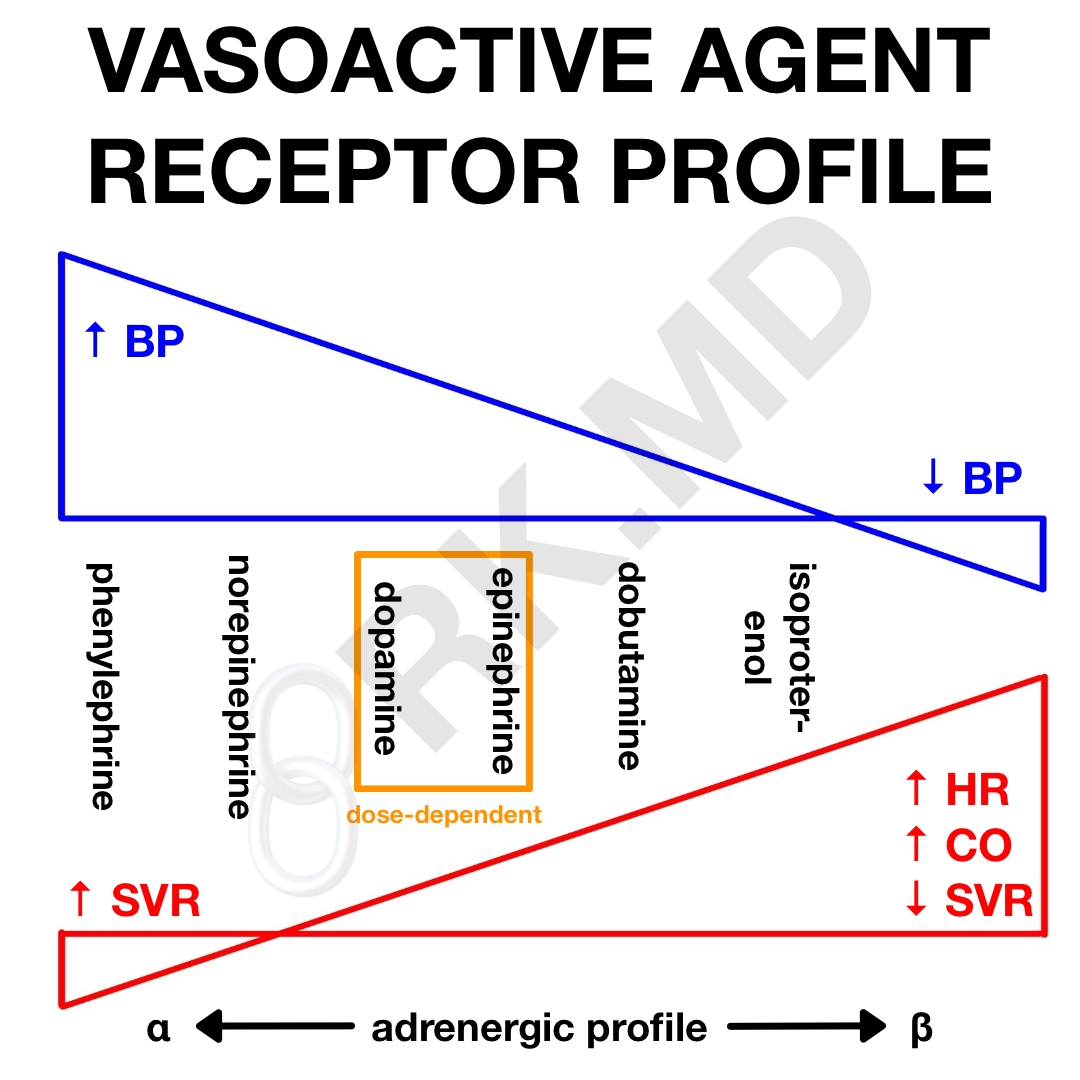
The literature contains so much ambiguity in terms of which drug is always better in certain situations, but there are fairly well-accepted indications for certain pressors (ie, norepinephrine as a first-line in septic shock). There are fewer examples of when/which vasoactive agents combine well to treat particular scenarios; however, this is a common practice in cardiothoracic anesthesia and the intensive care unit (ICU). How many times do patients come off cardiopulmonary bypass on milrinone and norepinephrine? What about all those septic shock patients on norepinephrine and vasopressin?
In my limited experience as a resident, I prefer agents the body makes naturally: epinephrine, norepinephrine, calcium and vasopressin being the big ones. Their normal, physiologic effects extend beyond the cardiovascular system to regulate glucose metabolism, the “stress response”, coagulation cascade, immunologic activation, etc.
Without going into too much detail, this image shows the general receptor profile of many common vasoactive agents. α-1 mediates systemic and pulmonary vasoconstriction to varying degrees. β-1 tends to augment chronotropy and inotropy. β-2 causes smooth muscle relaxation… like vasodilation in muscle beds. Keep in mind that agents like dopamine and epinephrine have a dose-dependent efficacy across multiple receptors.
Now how about some examples?
Right-ventricular (RV) failure is perhaps one of the most difficult situations to encounter in perioperative medicine, and is often due to pulmonary hypertension (pHTN). The RV is not accustomed to pumping against significant afterload created by the pulmonary vessels, so a dilated or sluggish RV on transesophageal echocardiography (TEE) may benefit from an inodilator like milrinone. This medication increases contractility and decreases pulmonary resistance improving RV output; however, it also decreases the systemic vascular resistance (SVR) creating hypotension which can affect coronary blood flow. Therefore, milrinone is often run with a afterload enhancer like norepinephrine (my preference) or epinephrine which also augments contractility but can create significant tachycardia and arrhythmogenicity.
Drugs like dobutamine and isoproterenol are primarily β-1/2 receptor agonists and are useful in low cardiac output states as they augment cardiac contractility and chronotropy while decreasing SVR – the net effect of improved forward flow. They’re both synthetic catecholamines which I’ve rarely seen used before any of the endogenous catecholamines.
Vasopressin is probably my go-to in a pinch. It uses a different mechanism to enhance SVR and tends to work when more traditional catecholamines fail (ie, severe acidosis). It also modulates systemic vasoconstriction more than pulmonary, so it’s an ideal choice in someone with RV strain or pHTN who is systemically vasodilated.
Outside of pediatrics, I’m not a fan of dopamine. Patients end up tachycardic with all sorts of ectopic beats. And there’s no evidence supporting its use for renal protection. 😛 Yeah, enough about that.
Now a quick blurb about receptor downregulation. In this example, if there’s a surplus of neurotransmitters and catecholamines (the substrates), then cells will often internalize receptors and regulate gene expression to limit the overall effect of the substrates. Feedback inhibition in action. 🙂
So by infusing a catecholamine, are we initiating the receptor downregulation cascade? Yes. Is it of clinical significance in the acute phase? Probably not. Nevertheless, receptor regulation (either down or up) is important to consider in chronic conditions like beta-blocker therapy and aging.
In conclusion, there’s no be-all, end-all combination of vasoactive agents for a situation. We must have a deep understanding of pathophysiology and pharmacology in the context of our patients’ comorbidities and current clinical condition to use in conjunction with monitors to determine the best options.
Drop me a comment with how you practice pressor management!






This is amazing, Rishi the MD!!!!
Thanks man!!
Thanks for the time saved for such a passionate topic…. In my limited time as an intern and my 11 months as a resident. Those are the protocols I’ve come across:
In the cardiac intensive care unit:
Only time i’ve used dopamine. 3-5 mcg/kg/min as a “diuretic” dose in patients with important RHF. Then with higher doses in AV block alongside isoprenaline sometimes…
In cardiogenic shock we used to start with dobutamine then associate to epinephrine in severe cases…
Then as a surgical ICU intern:
It was basically norepi/dobu in septic shock. ( we don’t have vasopressin in our country)
As I began my residency in a pediatric/obstetric ICU, most of our care is directed towards the postoperative care of cardiac surgery children….
I’ve seen all the cocktails in this context:
Epi combined with norepi and dobu
Epi combined with milrinone
I still gave to apprehend the reason behind these combinations…. Are they really useful? I still have to comb through the medical literature to find an article that really delves into these details….
Thanks again mate!!
Those are some fascinating protocols! Goes to show that more than one recipe works for the same clinical problem. 🙂