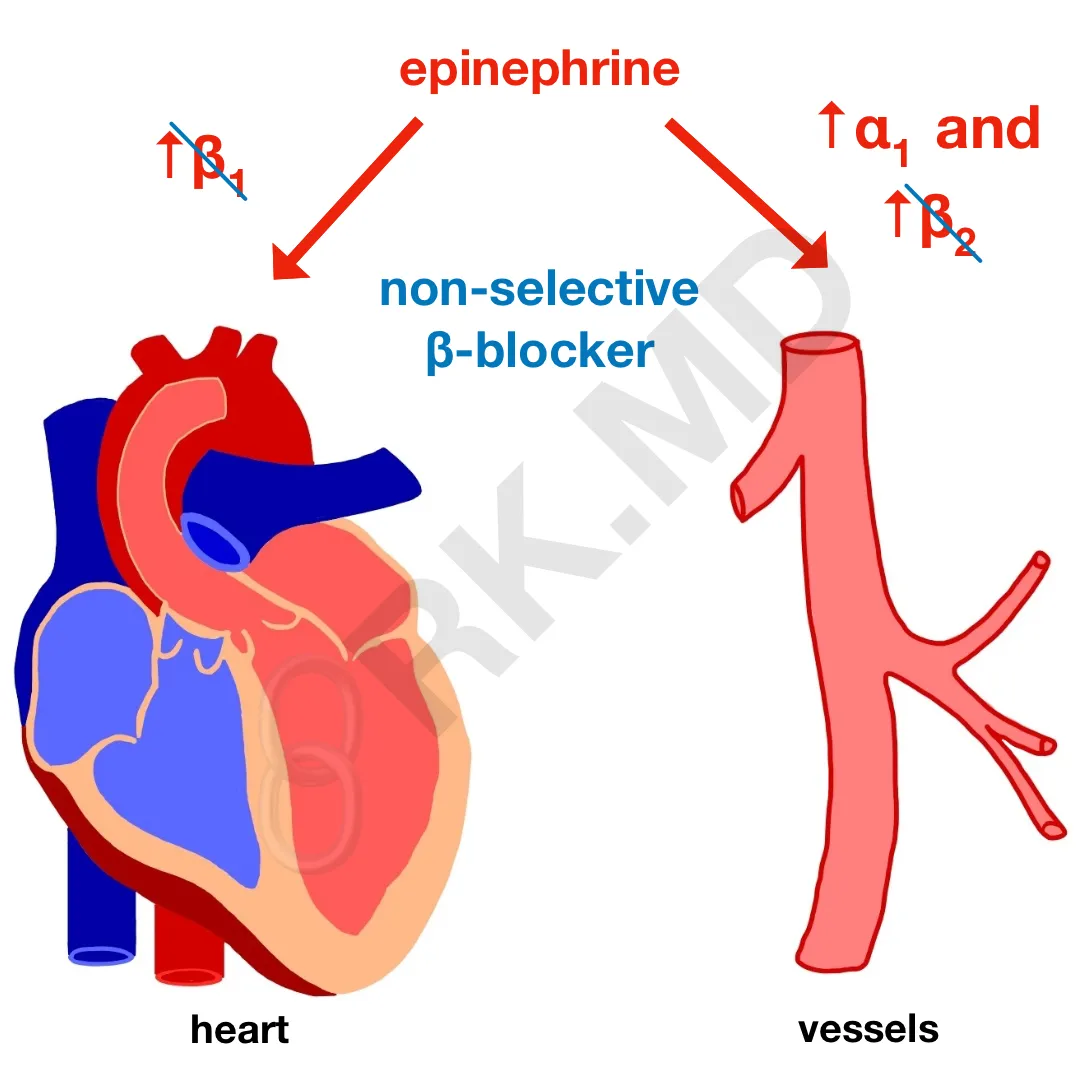
With the growing usage of β-blocker therapy to control everything from atrial fibrillation and congestive heart failure to migraine prophylaxis and essential tremor, we need to be more aware of potential pitfalls due to drug interactions. A classic example is the need for epinephrine in a patient on a non-selective β-blocker (ie, propranolol).
These non-selective agents block both β1 (inotropy, chronotropy) and β2 receptors (vasodilation). Yes, I’m disregarding non-cardiovascular implications of blocking β receptors to highlight the following point. From a pharmacologic standpoint, high doses of epinephrine will potentiate β1, β2, and alpha receptors. With a β-blocker like propranolol on board, epinephrine will primarily work through the α1 receptor – receptors on the peripheral vasculature involved with vasoconstriction.
The net effect will be unopposed vasoconstriction leading to significant increases in blood pressure. This hypertension will, in turn, activate pressure receptors in the carotid sinus to increase vagal tone on the heart leading to profound decreases in heart rate. Worst case scenario, the patient will become asystolic. Funny thing because in true asystole, the treatment is chest compressions and…. you guessed it… epinephrine! 😯
So why would we need to give epinephrine to these patients anyways? Anaphylaxis is one example! The mainstay of treatment will be epinephrine to stabilize further mast cell degranulation and promote bronchodilation, histaminergic blockers, steroids, and significant intravenous fluid resuscitation. A patient receiving β-blocker therapy may not mount the typical tachycardic response after receiving epinephrine. One will need to likely escalate their doses rapidly but be mindful of paradoxical bradycardia related to the mechanism above!
Another option is to give small doses of glucagon (1-3 mg followed by an infusion), but keep in mind that the evidence supporting the use of glucagon to reverse β-blocker therapy has only been shown in animal studies. Additionally, patients who receive this much glucagon invariably become nauseous and can vomit (oh the perils of an unprotected airway!)
Hopefully this makes sense, but if not, drop me a comment below with questions! 🙂






In a PEA/aystolic patient currently on beta blockers, is the 1mg of epi a high enough dose that you don’t typically need to consider giving glucagon to offset the beta blockade?
Although I don’t have data to answer this, I would most certainly say that 1 mg is PLENTY to overcome the effects of beta-blockade.
Thank you for this article. I have a mast cell disorder that causes tachycardia and spikes of high blood pressure. Propranolol has been one of the few drugs I can tolerate so far that helps. I have never had to use my epi, but anaphylaxis is common with my disorder. What would you advise in the event of anaphylaxis? Is there a better treatment protocol? I am always concerned about this. Thank you!
I don’t give medical recommendations over the Internet, so this is something definitely worth discussing with your allergist/immunologist. With that said, for true anaphylaxis, epinephrine is important to help stabilize mast cells from further degranulation.
But the response of epinephrine and activation of alpha and beta receptors depends on the concentration right.. the Dale vasomotor response shows initial surge followed by a drop because of beta2. Even if we block them , since dissociation occurs in the plasma, it shudnt affect much of the alpha effects as per my logic . Any thing missing in this deduction?
The general point of this post is to appreciate that certain medications (like epinephrine) might not elicit the expected response if patients are on medications that oppose their actions. Mileage will vary from person-to-person.
For a patient with severe sepsis, who had been receiving a non-selective beta blocker, would you consider alternative to levophed since it has similar properties to epi?
Not necessarily, but I wouldn’t be surprised if the patient didn’t respond predictably to pressors.
For patients on beta blockers, have you ever seen epinephrine cause reflex bradycardia or asystole in real life? I have not. Is there clinical experience or case reports to confirm this or is this a theoretical concern?
I have definitely seen reflexive bradycardia from small doses of epinephrine in beta blocked patients in the OR. I quickly switch over to agents like vasopressin in that instance.
Can you mention why you would want heart rate to increase when giving epi during anaphylaxis? I’ve read it’s to prevent cardiac collapse, but I’m not sure I completely understand why this would happen. Thank you so much for your great posts!
During anaphylaxis, a large amount of intravascular fluid is shifted into other spaces in addition to vasodilation. Together, this results in hypotension. Epinephrine not only helps stabilize mast cells but also augments cardiac output by increasing contractility and heart rate. In higher doses, it also improves vascular tone to mitigate the vasodilation. So yes, preventing hemodynamic collapse is indeed an important function of epinephrine (and large volume fluid resuscitation).
Is this related to the reason why bet blockers are contraindicated in patients who have cocaine on board?
Very similar reasoning! Except keep in mind that certain beta blockers with alpha blocking properties (like labetalol and carvedilol) are probably okay.