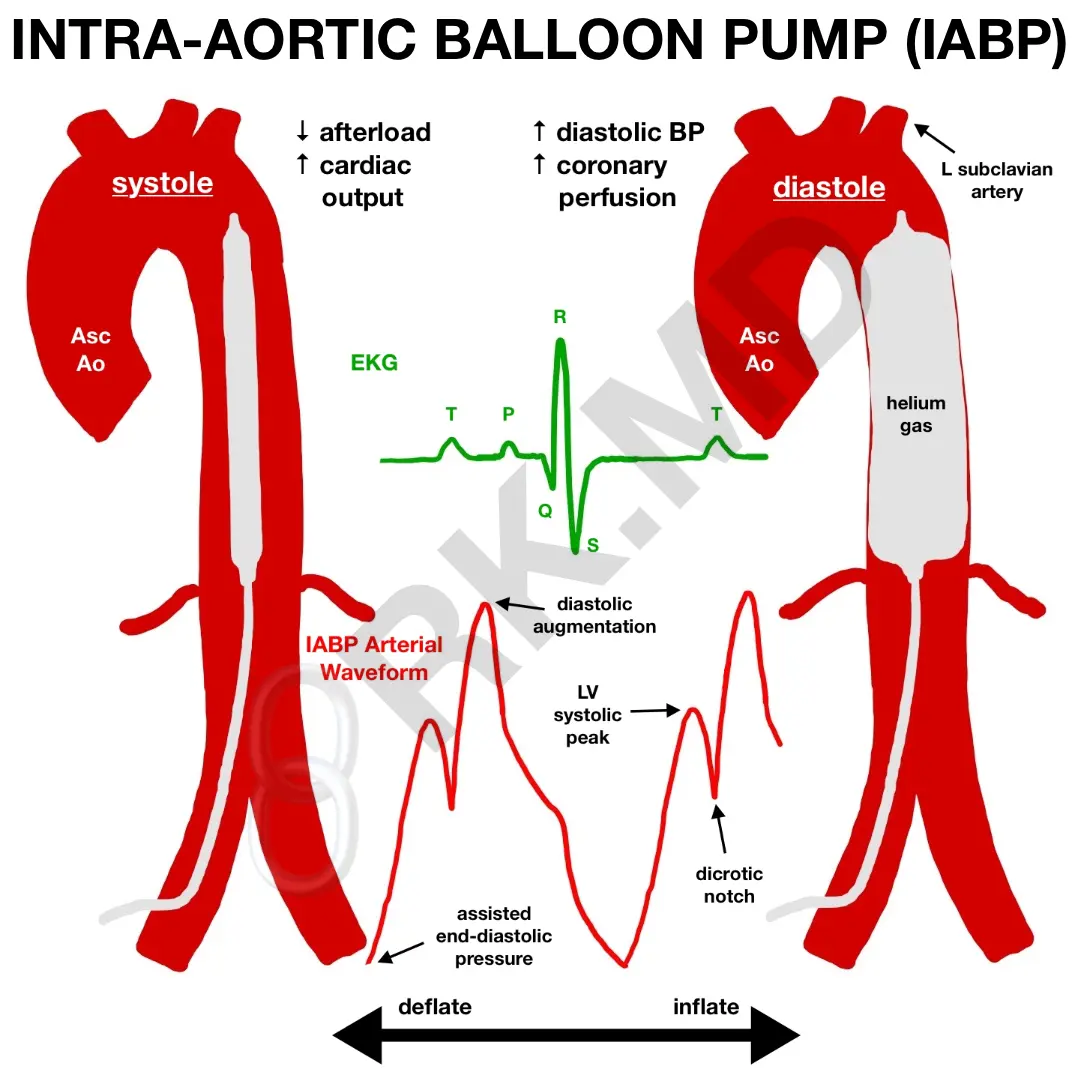
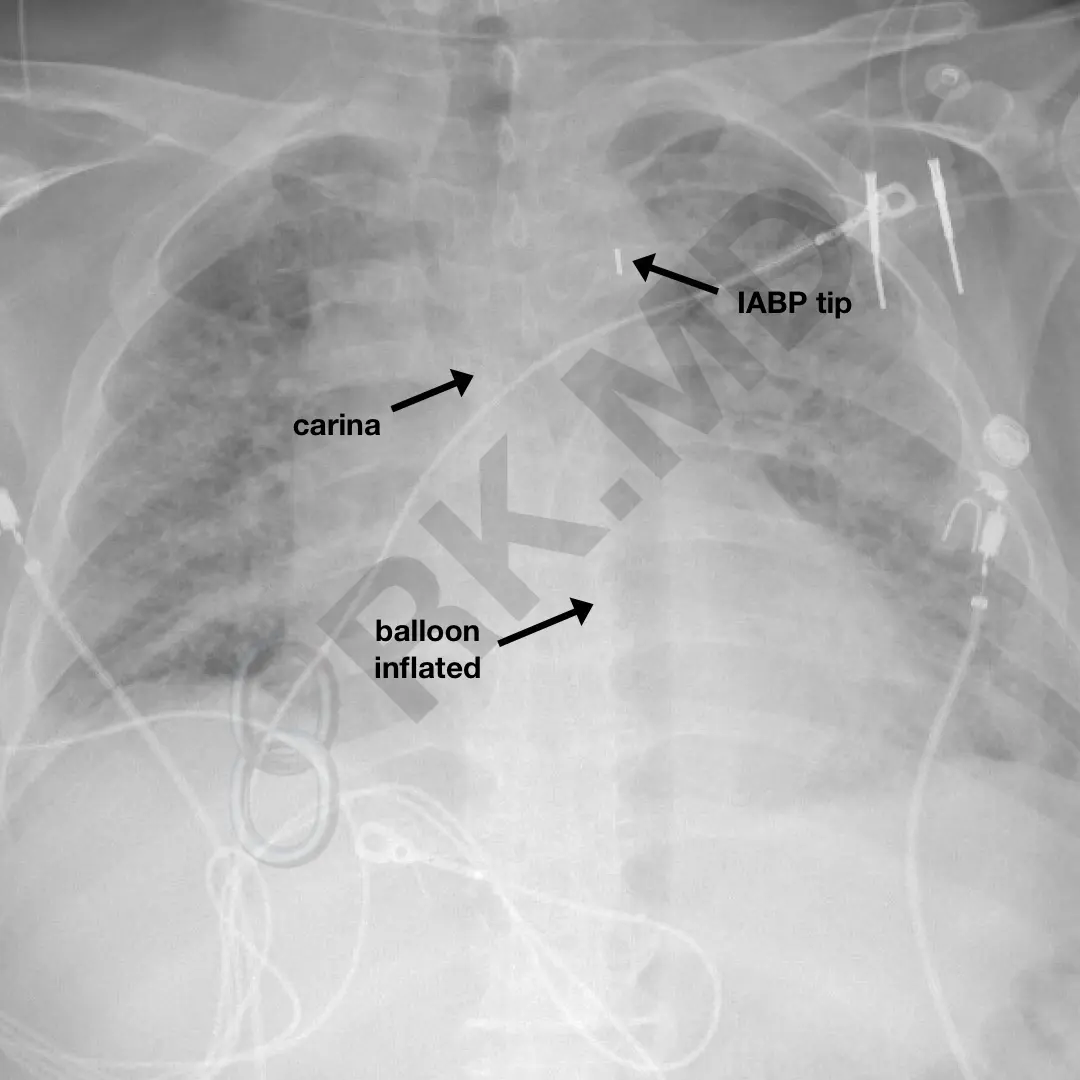
The intra-aortic balloon pump (IABP) is a form of mechanical circulatory support used in cardiogenic shock, facilitating separation from cardiopulmonary bypass, percutaneous coronary interventions, unstable angina, and thrombolytic therapy following myocardial infarction. The device is introduced from the femoral artery and guided into the aorta until its tip is distal to the left subclavian artery takeoff from the aortic arch. Therefore, when properly positioned, the IABP occupies the entire descending thoracic aorta and much of the suprarenal abdominal aorta. Although fluoroscopy/echocardiography can help guide placement, they are not necessary for emergent situations.
The basic premise behind the IABP is to increase aortic diastolic pressure to improve coronary perfusion. The balloon inflates rapidly with helium (less dense gas creates less turbulent flow and faster inflation/deflation) during diastole and creates an “augmented diastolic pressure” higher than normal. It is this diastolic pressure that is a primary determinant in coronary perfusion pressure. With the onset of systole, the balloon deflates, dropping the afterload and improving cardiac output. However, because of this constant mechanical agitation within the aorta, absolute contraindications include severe aortic insufficiency, aortic dissection, and severe peripheral vascular disease. In general, the sicker the left ventricle, the less the IABP can actually help compared to devices like the Impella.
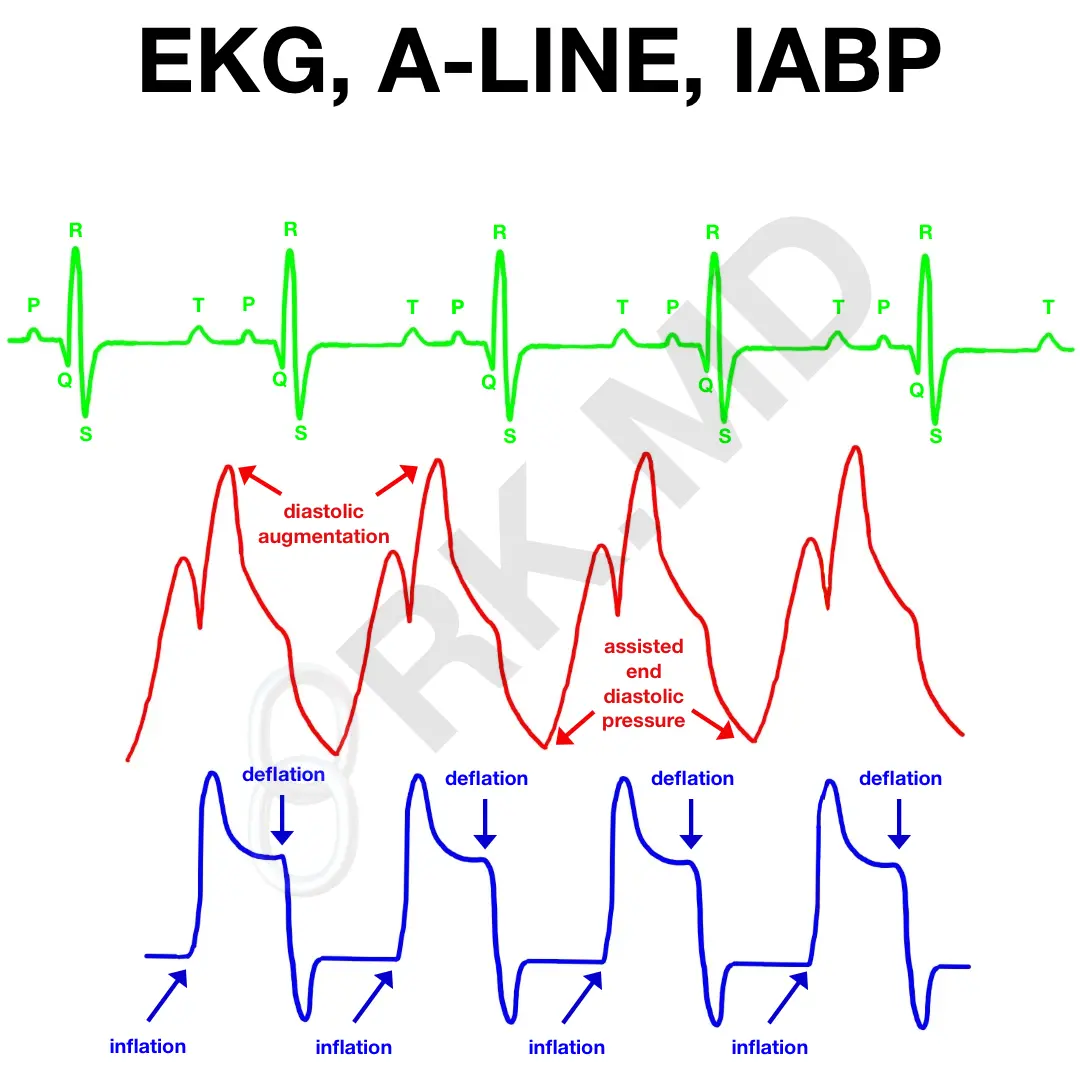
So how can the IABP tell when systole and diastole occur, and in turn, know when to deflate and inflate? We can trigger the IABP by syncing it with the patient’s EKG, the arterial waveform (the dicrotic notch represents aortic valve closure and the end of systole), and even pacing spikes. We can even set the IABP to augment every cardiac cycle (1:1), every other cycle (1:2), etc.
Here’s an illustration I sketched how the EKG, arterial line (A-line), and IABP waveforms line up vertically. The IABP will inflate at the end of systole (where the dicrotic notch would appear on the arterial waveform) and in the mid-T wave on EKG. Then, it deflates on the R wave.
It’s imperative to check that the IABP is firing appropriately relative to the cardiac cycle. Having the balloon inflated during systole will cause a significant impediment to forward flow, and having it deflated during diastole defeats the purpose.
These are just the basics of this useful counterpulsation device. Please drop me a comment below with questions! 🙂






Thank a bundles
Long time follower here. Was wondering what resources you’ve used to learn everything you can about IABP. Outside of Balooki book and manufacturer manuals, what else have you used to learn about the IABP in detail?
Hey Landon! I saw your email and you already covered some of the resources I’d recommend (like the manufacturer manual). Additional material can be found on PubMed like this StatPearls with some more physics here.
Hi I was wondering how we can measure AUG and Afterload reduction, when we switch to 1:2?
Thank you
Hello Dr. Rishi,
First and foremost great site I actually just found it. As a critical care nurse I’ve cared for patient on iabp and Impella.
I do have some questions;
1) How does the MAP displayed on the iabp console gets calculated?
2) If a patient is 100% paced do you switch to pacer mode on iabp or keep on auto? Does it really matter if left on auto?
3) when do you use the asynchronous trigger during open heart surgery and what’s the rationale for using it ?
Thank you ahead of time for everything you do Dr. Rishi
Hello Emel! Thanks for the questions!
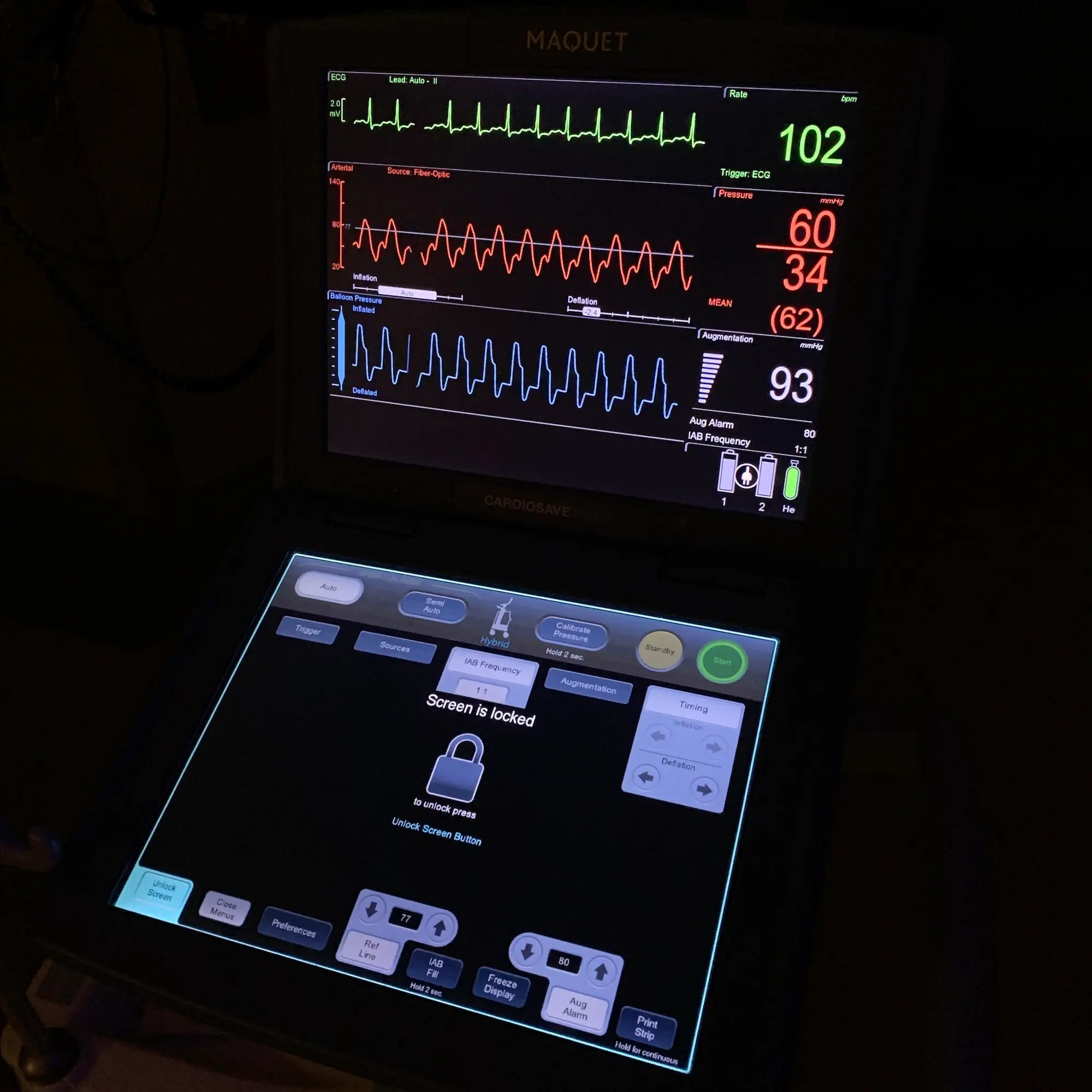
1.) The IABP can tell the difference between an unassisted SBP, augmented DBP, and end diastolic pressure. If you look at the example I have in the post, the IABP console is displaying an SBP of 60 mmHg and DBP of 34 mmHg. This would be a MAP of 42 mmHg if you calculate it out; however the listed MAP is 62 mmHg because this is the effective mean pressure factoring in augmentation. If the patient has a peripheral arterial line (femoral, radial, etc.), the MAPs should correlate between the IABP and A-line, but the SBP/DBP can be very different.
2.) If a patient is intermittently paced, I’ll just keep the IABP on auto. It’s the trigger I use the overwhelming majority of the time. If someone is truly 100% pacer dependent, then I’ll use a pacer mode.
3.) My surgical colleagues leave the IABP off during cardiopulmonary bypass, but there’s some literature suggesting improved overall systemic perfusion with “pulsatility” created by an IABP.
Thank you so much for this helpful post – the way you provide information is so straightforward and easy to understand. Thank you for taking the time to do this 🙂
Thank you for the kind feedback, Kayla! I really appreciate it! 🙂
How does the balloon deflating during (or just prior to) systole decrease your afterload?
You have a sudden increase in the volume of the aorta just distal to the heart because the space-occupying balloon has rapidly deflated. Negating the effects of aortic compliance and assuming a fixed temperature, this will create almost a suction-like effect to help transiently reduce afterload.
Hi Rishi,
Amazing article. Thank you! I know we are talking liquid and not gas here, but does this volume and (presumed) pressure change creating the suction relate in any way to Boyle’s Law, or is it altogether a different mechanism?
Thanks again!
Andy
I actually DO think about it this way in the sense that while holding temperature constant, when the IABP deflates, the aortic “volume” has increased thereby instantaneously decreasing pressure.
hi, wow! love your site. what happens if a patient is in AF or otherwise irregular heart rhythm? how does the IABP work or adjust to that? (or does it / can it?)
It can still be synchronized to the heart rhythm via the arterial line waveform. 🙂
What if there is no arterial waveform?
There’s an internal trigger mode.
When reviewing doctor’s notes after my husband’s heart attack and emergency 6x CABG – I wanted to know what IABP was – Your site is AMAZING! Even I could grasp enough to know what happened and why. THANK YOU!
I’m so glad you found it useful, Lukie! As you can imagine, writing about these topics in a way that’s both simple yet detailed is a difficult task. I’m glad you find the site informative! Best wishes to you and your husband! 🙂
Thank you for the great info. Would you explain during a flight and changes in atmospheric pressure how to adjust/troubleshoot the IABP?
Hey Mira! I’m actually not sure!
How quickly after fluid, pressors, inotropes does one after cardiac surgery go to IABP? What if the patient’s CO is still crummy? How does one adjust the IABP to maximize its usage in this case?
That’s a tough question. Sometimes IABP is placed pre-emptively and other times it’s placed just to augment coronary perfusion by mechanical means in conjunction with pharmacologic therapy. There’s not necessarily a specific number I’m chasing, and most of the heart failure surgeons I work with are very aggressive about utilizing mechanical circulatory support (MCS) sooner-than-later which I’m honestly a fan of. There are plenty of MCS options to consider too (IABP, Impella, etc.) depending on the goals and hemodynamic situation. If I’m using a IABP on “maximum” settings, it’ll be fully augmenting every beat (1:1, 100% augmentation).
First of all, love your site, it’s brilliant. I have a question about if a patient has an open chest with IABP and requires internal cardiac massage, what setting should the iabp be on?
Excellent question, Kim! My personal bias is to ensure the IABP is switched (if not already on) an arterial line waveform trigger. The concern would be dyssynchrony between your compressions and the inflation/deflation of the balloon. If it’s on the arterial waveform trigger, the balloon should sync up based on adequate chest compressions generating an arterial upstroke. 🙂
Thanks for the quick response, during a code I have never thought twice about switching to pressure, I just never thought of an open chest code in relation to the iabp.