Pressure transducers are a cornerstone for measuring arterial, central venous, pulmonary artery, and even intracranial pressures. These transducers operate through a Wheatstone bridge, a circuit with an arrangement of resistors of known resistance except for one. This unknown resistor is a strain gauge coupled to your pressure of interest (A-line, CVP, etc.) via incompressible tubing usually filled with (heparinized) saline. As the force on the gauge changes due to fluctuations in pressure, the intrinsic resistance within the gauge also changes, causing a shift in the current flowing through the Wheatstone circuit. An algorithm is used to interpret this flux in current as a change in the pressure magnitude – this is the number we ultimately read on the monitor. 🙂
The waveform produced by the monitor is dependent on what is being measured. For example, the morphology of a CVP tracing looks very different than that of an arterial line. For this post, I’ll talk about arterial lines since they’re fairly common in the intensive care unit (ICU) and operating room (OR).
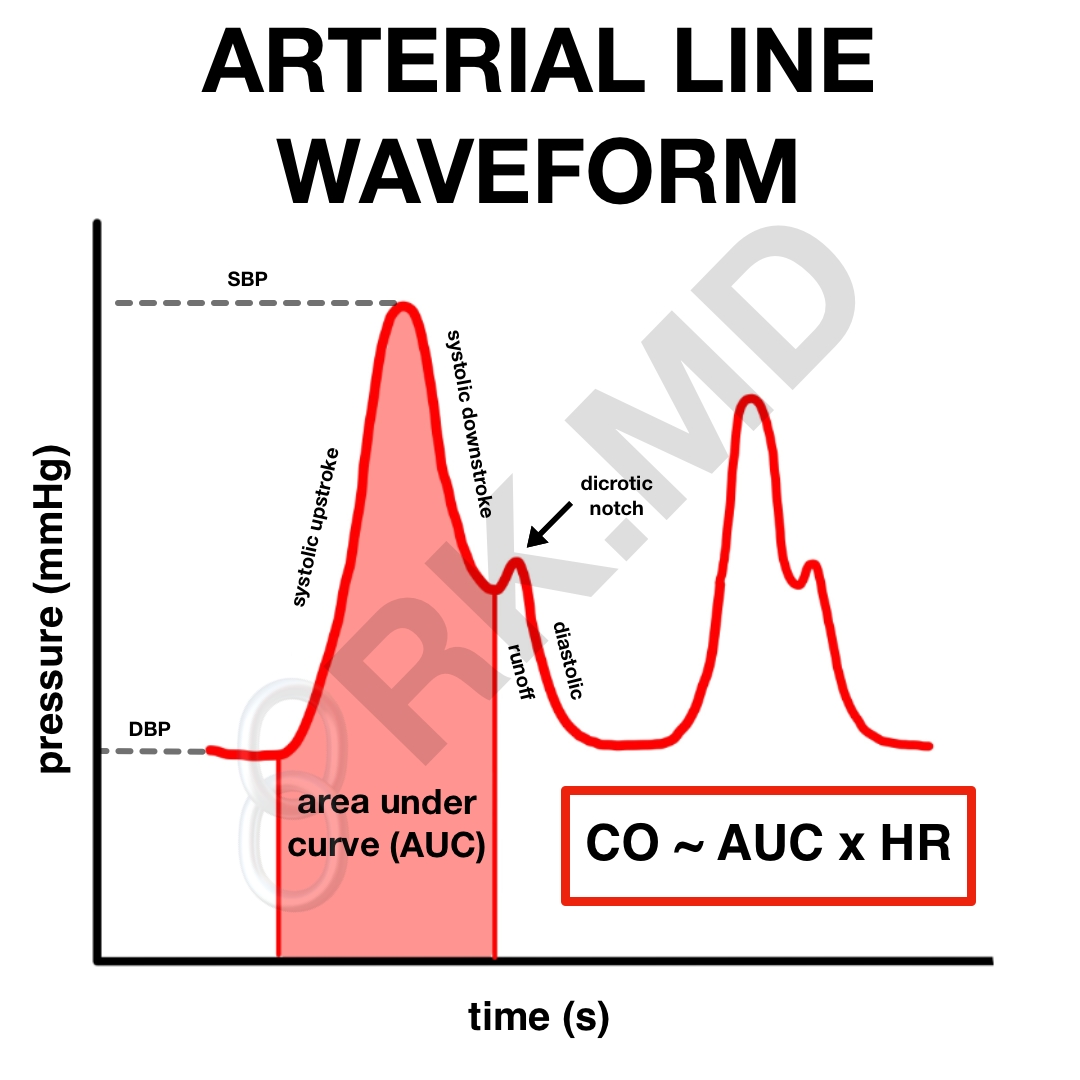
First, unlike an automated blood pressure cuff, an arterial line (A-line) directly measures the systolic, diastolic, and mean arterial pressure. In the waveform above, the nadir corresponds to the diastolic blood pressure (DBP). The upstroke immediately after represents ventricular contraction and culminates at the systolic peak, which represents the systolic blood pressure (SBP).
As the ventricle relaxes, its pressure falls below that of the aorta resulting in the closure of the aortic valve, which creates a reflected pressure wave represented by the dicrotic notch. As one moves distally in the body (think about an A-line in the foot versus in the radial artery), the dicrotic notch will appear more delayed. Also, the dicrotic notch will occur at much lower pressure in severely vasodilated patients (septic shock, neurogenic shock). In this case, you may want to consider afterload enhancement with agents like norepinephrine and/or vasopressin.
Now how does the overall morphology of the arterial line change as you move more distally? Again, let’s consider a patient with two arterial lines: one in the radial artery and one in the dorsalis pedis artery (foot). The mean arterial pressure (MAP) between the two should be similar, but the more distal foot A-line will have a higher SBP and lower DBP than the radial A-line. This results from reflected pressure waves at all the branch points blood encounters as it exits the heart and barrels down the arterial tree.
Drop me a line with questions below! I’ll be addending this post with more information in the coming days.






Hi Dr. Kumar,
Firstly, thank you for all of your FOAMed–it has proven invaluable to me. Secondly, based on this graphic, is there a way to use that AUROC to actually approximate C.O. at the bedside?
Thank you again, happy holidays!
Thanks for the kind words, Cory! Honestly, from the perspective of a clinician, I don’t think this is necessarily accurate nor practical. If you “eyeball” the area under the arterial line curve as illustrated in the diagram, sure, it might correlate to the cardiac output, but good luck trying to assess this if the patient is tachycardic (without adjusting the monitor’s sweep speed), the arterial line signal is inconsistent (e.g., due to patient movement), etc. I leave these calculations to monitoring systems like FloTrac.
If I’m trying actually to calculate cardiac output at the bedside, I’ll use bedside ultrasound and determine the outflow through the aortic valve.
What does it mean by optimum waveform on cardiac monitor scale? What scale we should pick? If we click optimum waveform how would this affect actual blood pressure?
Adjusting the scale doesn’t affect the actual blood pressure, but it makes it easier to appreciate the morphology of the waveform. I want a scale which approximates the patient’s actual blood pressure. For example, I’m not using a scale of 200 mmHg if my patient’s systolic pressure is in the 100 – 130 mmHg range.
can we apriciate volemic status due to arterial waveforms?
Hi Rishi,
Km reviewing the Sheehy’s emergency text and came to your website for more explanation on the arterial waveforms. The text notes an anacrotic notch and volume displacement curve before the diastolic runoff. Are these necessary components to understand for emergency care? Thank you
Hey Ellie! Honestly, we can complicate monitors to no end. The short answer is no – if you understand the arterial upstroke, dicrotic notch, and overdampening/underdampening, I think you’re set!
Hey Rishi! Now that I’m a new grad in the ICU, I find myself referencing your blog often and was very pleased to find that you had exactly what I was looking for… A line waveforms! This was very helpful as well as the response to the comments. I also love your pictorial supplementation to your explanation. Thanks for all you do!
Glad you found it helpful! 🙂
Hi Rishi,
Would it be ok if I reproduce your picture of arterial waveform in an educational pack for Critical Care nurses please. I will of course reference the article.
Many thanks
Hey Karen! That’d be such a privilege! Thanks for letting me know! 🙂
Hey! I have had issues with trouble shooting art lines. If I am having a hard time drawing blood off the art line what are things I can do for trouble shooting purposes.
We’re often called for arterial lines that “transduce but don’t draw back”, and honestly, if the patient still warrants having an arterial line, I’ll just replace it either by rewiring the existing line or placing an entirely new one. Usually this is due to a kink in the catheter, but can also be from vasospasm/vasoconstriction of the vessel more proximally.
Could you please elaborate on reflected pressure waves and why is distal SBP is higher and DBP is lower than proximal readings from radial a-line. Thank you!
Great question! With each heart beat, a pressure wave is transmitted down the arterial tree with the ejected stroke volume. As blood travels further from the heart, it encounters more branch points and narrower blood vessels. This translates to more resistance (especially in older patients with “stiff” arteries) causing some of the pressure wave to be reflected backwards (a pathway with less resistance).
Now why does the systolic increase? I think it’s partly due to the acceleration of pressure waves through the narrower vessels. The diastolic pressure decreases because a component of the initial pressure wave is now reflected retrograde and essentially subtracted from the diastolic pressure you would expect.
Fluid dynamics. 😯
Thank you for the reply!
So in terms of numbers I would see 120/80 in R a-line and 140/60 in fem a-line and that essentially would be the same since MAP should be equal. And that would prompt me not to chase higher SBP in fem line considering the fluid dynamics? ?
So the problem is yes, from a purely academic standpoint, we should probably focus more on MAP rather than SBP. However, a lot of physicians write for blood pressure goals and PRN anti-hypertensives in terms of SBP which is affected by where the arterial line is located. I’ve tried explaining this to colleagues with varying levels of success.
You mention there is no need to replace the vented stopcock caps with non-vented ones. I’m wondering why that is, as I believe it is taught to always replace with caps without the holes (I’ve always assumed for infection prevention?). Thanks for all you do!
Hey Allie! Maybe it’s just an institutional thing (and depends on the setting). In the OR, I never changed the stopcock caps to non-vented ones, but it sounds like in some ICUs, this is the norm? Goes to show the variability in even the smallest things from place to place, haha.