Ventricular assist devices (VADs) can support the right (RVAD) and left (LVAD) ventricles and were initially utilized as mechanical circulatory support devices for bridge-to-transplant (BTT) in heart failure. Modern continuous flow VADs are also being used for destination therapy (DT) for those who are not candidates for cardiac transplantation or are too ill to wait for a donor. The options available for advanced heart failure therapy are growing each year, so we are increasingly likely to encounter patients with VADs.
When I started as an attending in late 2019, there were two VAD options: Medtronic’s HeartWare HVAD and Abbott’s HeartMate 3 (HM3). The HVAD was taken off the market in summer 2021 after “a growing body of observational clinical comparisons indicating a higher frequency of neurological adverse events, including stroke, and mortality with the HVAD System as compared to other circulatory support devices available to patients.” As such, I’ll focus only on the HM3 in this article.
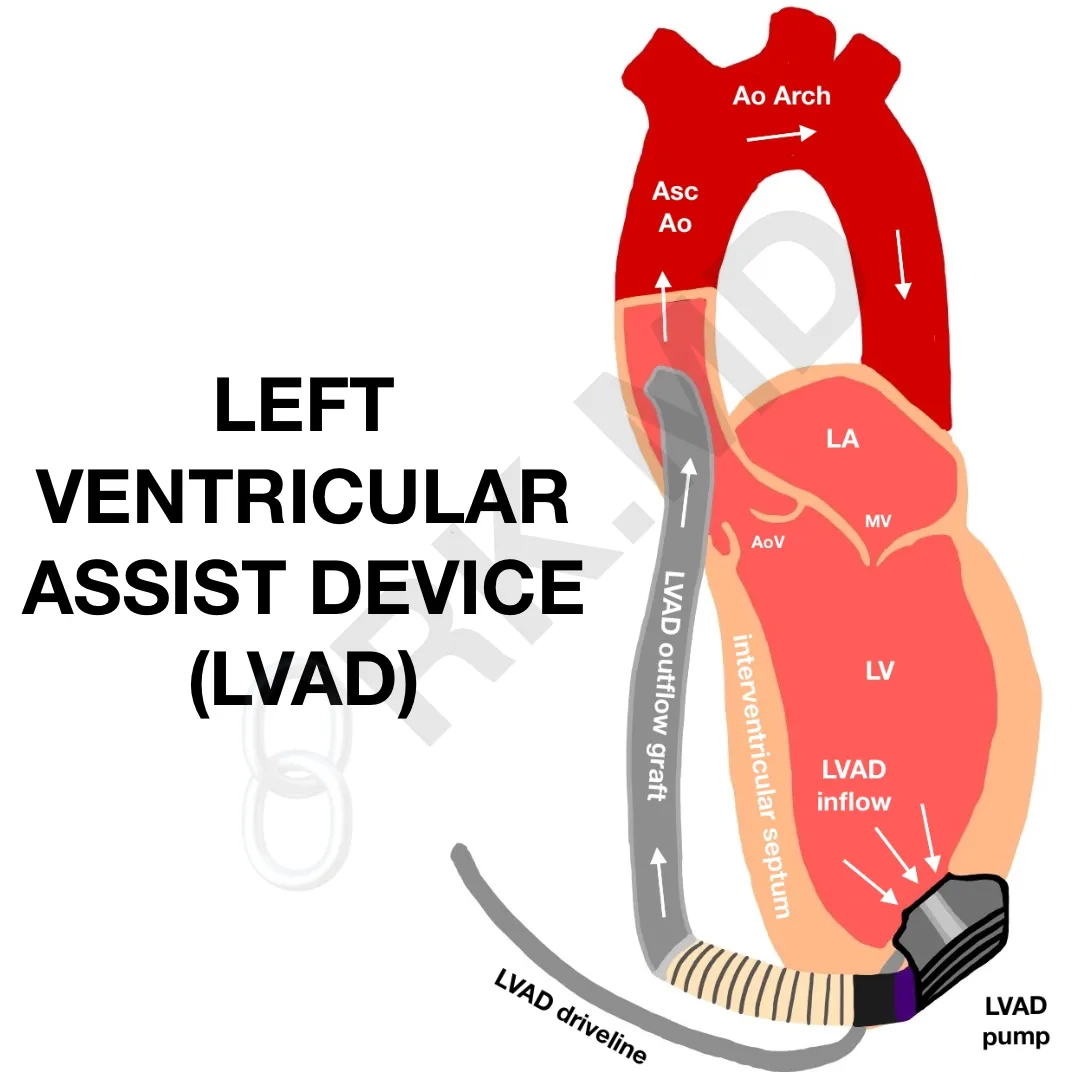
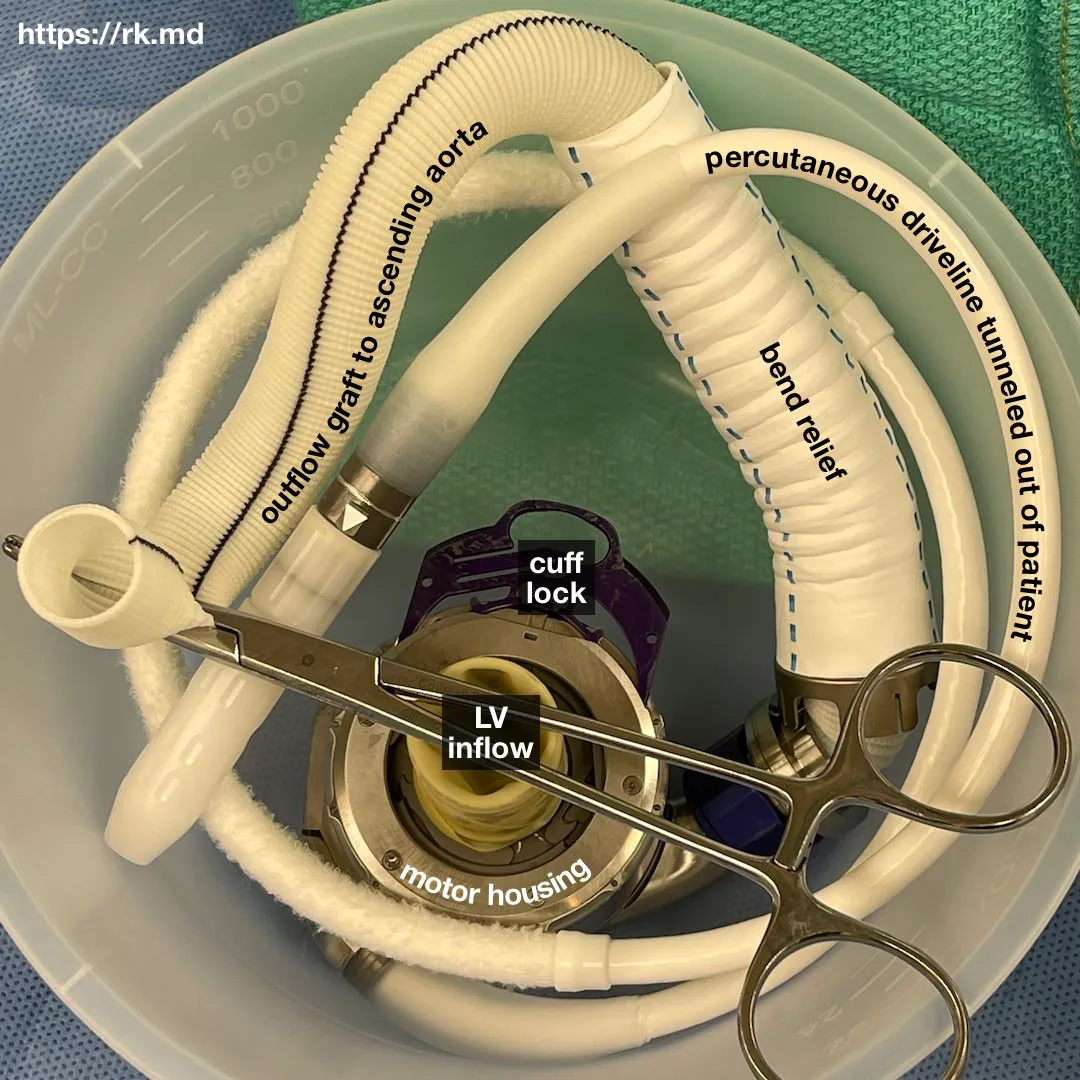
The HM3 has a motor that spins 2,000-5,500 times per minute! This rotary action propels blood flow from the inflow cannula in the left ventricular apex to the outflow cannula connected to the proximal aorta. Because of the constant flow, patients may not have a palpable pulse depending on their underlying ventricular function and degree of left ventricular loading. This is in line with the Frank-Starling Law of the heart.
LVADs assist the LV by promoting forward systemic flow. It’s important to keep right heart failure in mind and consider pulmonary vasodilators with inotropic support of the right ventricle (RV) if needed. Remember, a significant portion of RV systolic function hinges on LV function. If the LV does not have to do much work anymore because of the LVAD, the RV could fail.
Pump speed (as RPM) is the only variable programmed by an operator, but a VAD machine will display other parameters:
- Power – basically the energy needed to create flow. An acute increase in power requirements could be related to thrombosis within the LVAD and warrants additional anticoagulation and/or thrombolytics consideration.
- Flow – a surrogate for the cardiac output. For a fixed RPM, a higher flow rate implies decreased resistance, as in sepsis or vasodilation. A lower flow suggests decreased intravascular volume (i.e., hypovolemia or bleeding). Flow is related to the power, pump speed, patient’s blood viscosity, and pressure differential across the pump.
- Pulsatility index (PI) – represents the balance of native ventricular function and unloading by the continuous flow VAD. An increase could suggest recovery of ventricular function, whereas a decrease represents hypovolemia, worsening ventricular function, or increased pump speed causing more ventricular unloading and less pulsatility.
As a cardiothoracic anesthesiologist, I’m routinely involved with LVAD implantation surgery, LVAD pump exchanges, etc. Transesophageal echocardiography (TEE) allows me to determine how the rest of the heart is reacting to the LVAD, titrate my vasopressors/inotropes accordingly, and help gauge the pump speed necessary to optimize LVAD outflow without compromising ventricular filling to cause suction events. Here’s an example of a midesophageal 4-chamber TEE view showing a well-placed LVAD inflow.
We also have to consider other things in patients requiring VAD support:
- These patients require anticoagulation and are pre-disposed to clot formation due to stasis within the native heart, especially around the aortic valve. Therefore, lookout for signs of embolic phenomena.
- Dysrhythmias are often caused by the inflow cannula irritating the ventricular septum. Fluid resuscitation and slowing the pump speed can allow the ventricle to fill more and alleviate this suction effect.
- Aortic insufficiency (“regurgitation”) is fairly common the longer VADs stay in, and this can actually create a loop within the VAD. Left ventricle → VAD inflow → VAD outflow → aorta → back into the left ventricle through an incompetent aortic valve.
- Hemolysis from physical agitation of red blood cells making contact with the rotor pump. Monitor carefully whenever the pump speed is adjusted.
- Infections can occur in the VAD driveline or the pocket housing the pump (depending on the VAD). Typical organisms include Staphylococcus, Enterococcus, and Pseudomonas. Empiric antibiotics should be initiated, and sometimes surgical debridement is required for deeper wound infections.
- Arrest: In the event of cardiopulmonary arrest, if the VAD is still functional, typically, a “chemical code” is warranted after investigation of the VAD and possible causes of, for example, hypotension. If the VAD function has truly stopped or is not functioning appropriately with resulting hypotension, external chest compressions are recommended.







Can you explain suctions events vs PI events? I understand hypovolemia is a cause but not always. What would you see change on your monitor setting with each and how can you tell the differences? Thank you
I don’t know what you mean by a “PI event?” A drop in PI? Suction events can occur when the LVAD inflow is reduced due to a decreased cavity size (pump speed is too high, LV hypovolemia, etc.) causing obstruction of the inflow from the adjacent LV muscle (e.g., papillary muscle, interventricular septum).
A drop in PI is typically due to decreased volume in the LV (systemic hypovolemia, acute RV dysfunction, etc.) or a decrease in native LV contractility.
It’s not just about what’s on the monitors – understanding the pre-test probability of these things occurring is important. These can both present as hypotension and cause decreased flow on the LVAD console, so diagnostics (echocardiography) and testing the response to therapy (reducing LVAD pump speed, small bolus, inotropic support) can help with understanding the physiology at play.
Thank you so much for your response! I think the term PI event gets thrown around at my work place referring to a drastic change in the PI value (average PI being different for everyone) but when it is cut in half or doubled with no change in speed or flow. From my understand a suction event will always change your flow and speed along with the PI on the monitor. Maybe this is just a term used on my unit?
My unit looks at PI events as well and we are trained to document them and whether they included a speed drop. It shows on our LVAD wall units. Our providers typically only want to be notified of one if there is a speed drop.